
This reaction can give a very pure product, e.g. The resulting hydrogen chloride gas is absorbed in deionized water, resulting in chemically pure hydrochloric acid. The pure chlorine gas can be combined with hydrogen to produce hydrogen chloride in the presence of UV light.Īs the reaction is exothermic, the installation is called an HCl oven or HCl burner. In the chlor-alkali industry, brine (mixture of sodium chloride and water) solution is electrolyzed producing chlorine (Cl 2), sodium hydroxide, and hydrogen (H 2). Most hydrogen chloride produced on an industrial scale is used for hydrochloric acid production.ĭirect synthesis File:Hydrochloric Acid Burner Flame.ogg While the spring constants are very similar, the reduced masses are different causing significant differences in the rotational energy, thus doublets are observed on close inspection of each absorption line, weighted in the same ratio of 3:1. Naturally abundant chlorine consists of two isotopes, 35Cl and 37Cl, in a ratio of approximately 3:1. However, the vibrational energy of HCl molecule places its absorptions within the infrared region, allowing a spectrum showing the rovibrational modes of this molecule to be easily collected using an ordinary infrared spectrometer with a conventional gas cell. The value of B is much smaller than ν e, such that a much smaller amount of energy is required to rotate the molecule for a typical molecule, this lies within the microwave region. selection rules state that ΔJ is only able to take values of ± 1. They are characterized by the rotational quantum number J = 0, 1, 2, 3. Because of quantum mechanical selection rules, only certain rotational modes are permitted. Instead, two sets of signals (P- and R-branches) are seen owing to rotation of the molecules. This absorption corresponding to the Q-branch is not observed due to it being forbidden by symmetry. To promote an HCl molecule to the v = 1 state, we would expect to see an infrared absorption about 2880 cm −1. At room temperature, almost all molecules are in the ground vibrational state v = 0. The infrared spectrum of gaseous hydrogen chloride, shown below, consists of a number of sharp absorption lines grouped around 2886 cm −1 (wavelength ~3.47 µm). One doublet in the IR spectrum resulting from the isotopic composition of chlorine. Infrared (IR) absorption spectrum File:HCl37.JPG 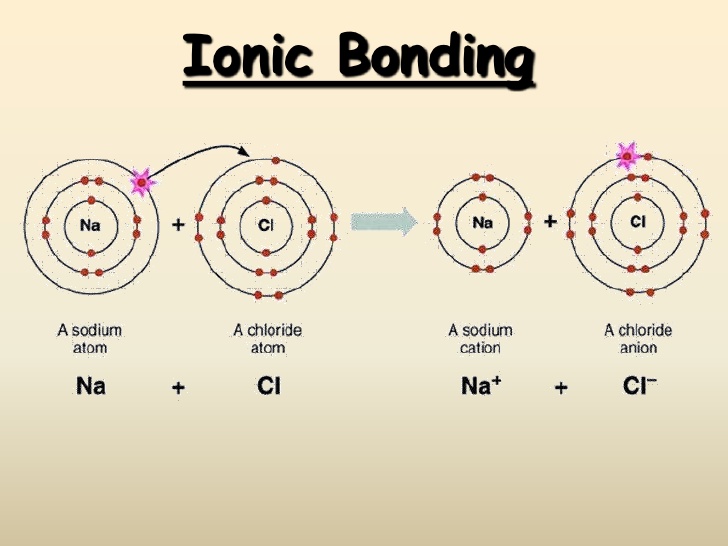
Solubility of HCl (g/L) in common solvents Temperature (☌) Analysis of spectroscopic and dielectric data, and determination of the structure of DCl (deuterium chloride) indicates that HCl forms zigzag chains in the solid, as does HF (see figure on right). However, the hydrogen atoms could not be located. In both structures the chlorine atoms are in a face-centered array. X-ray powder diffraction of the frozen material shows that the material changes from an orthorhombic structure to a cubic one during this transition. For example, hydrogen chloride can dissolve in certain other solvents such as methanol, protonate molecules or ions, and serve as an acid- catalyst for chemical reactions where anhydrous (water-free) conditions are desired.īecause of its acidic nature, hydrogen chloride is corrosive, particularly in the presence of moisture.įrozen HCl undergoes phase transition at 98.4 K. Even in the absence of water, hydrogen chloride can still act as an acid. 
The acid dissociation or ionization constant, K a, is large, which means HCl dissociates or ionizes practically completely in water. The resulting solution is called hydrochloric acid and is a strong acid. Upon contact, H 2O and HCl combine to form hydronium cations H 3O + and chloride anions Cl – through a reversible chemical reaction: In part because of its high polarity, HCl is very soluble in water (and in other polar solvents). Consequently, the molecule has a large dipole moment with a negative partial charge δ – at the chlorine atom and a positive partial charge δ + at the hydrogen atom. Since the chlorine atom is much more electronegative than the hydrogen atom, the covalent bond between the two atoms is quite polar. Hydrogen chloride is a diatomic molecule, consisting of a hydrogen atom H and a chlorine atom Cl connected by a covalent single bond. Hydrochloric acid fumes turning pH paper red showing that the fumes are acidic
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
